Open Positions
Product Sales Specialist
General: Full-Time, Remote or On-site (Oregon)
Job Category: Sales
Base Salary: $75,000- $85,000
Total compensation: Expected $185,000+ dependent on performance.
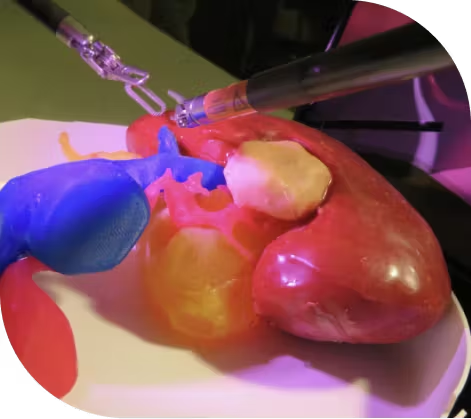
Overview of Lazarus 3D: Surgeon skill and experience are the biggest determinants of patient outcomes. Unfortunately, surgeons learn by operating on patients and sometimes they make costly mistakes. Lazarus 3D’s Pre-Sure platform allows surgeons to rehearse every surgery ahead of time on a replica of their patients’ organs. This unparalleled opportunity to exercise their surgical plan on Pre-Sure is game-changing in surgical care as it can transform patient safety and outcomes. Lazarus 3D’s Skill-Sure platform delivers world-class solutions to healthcare professionals interested in reducing the learning curve, innovation and adoption of new medical devices. Pre-Sure and Skill-Sure models mimic soft, realistic textures of real organs and provide a unique hands-on operative experience.
Our mission is to help doctors operate with confidence: Lazarus 3D is driven by a singular mission: to enhance patient safety and outcomes through tailored solutions that empower surgeons to operate with unwavering confidence. Our vision is ambitious yet resolute—to eradicate surgical errors, ensuring that every patient receives the best care possible.
Join us at Lazarus 3D as we redefine the boundaries of surgical precision and pave the way for a future where impeccable care is the standard.
Introduction: We are currently seeking a passionate, experienced and strategic medical device visionary to join our team as the Product Sales Specialist for the US Market. This role will be ultimately responsible for the business success of Lazarus 3D’s products, namely Pre-Sure and Skill-Sure. Primary clients include health systems, private practices, medical device companies and residency/fellowship programs.
Your Role at Lazarus 3D: Lazarus 3D is seeking a full-time Product Sales Specialist position for Pre-Sure and Skill-Sure. This is an opportunity to get in on the ground floor of a rapidly expanding startup with a mission to improve patient safety and outcomes in surgical care. Established in 2016, Lazarus 3D’s innovative solutions provide healthcare professionals with tools that improve their surgical skills, reduce the learning and adoption curve, and help them operate with confidence. Lazarus 3D’s turn-key, custom-tailored solutions support and promote medical innovation for medical device manufacturers through Skill-Sure, providing medical device teams everything they need to train and innovate. Lazarus 3D’s flagship product line, Pre-Sure, is the first and only FDA-cleared diagnostic patient-specific tool that allows surgeons to rehearse their upcoming surgeries ahead of the curve. Pre-Sure provides an unparalleled opportunity for surgeons to exercise their surgical plan and perfect it before they operate, driving improved safety, efficiency, and outcomes for patients globally.
As a Product Sales Specialist, you take pride in representing our Pre-Sure and Skill-Sure divisions at Lazarus 3D. Your keen understanding of our diverse portfolio of surgical training and rehearsal products and belief in the value and quality they provide to surgeons and patients fuels your confidence. Our customers trust you and appreciate your knowledge and curiosity when seeking customized solutions to meet their needs. You enjoy travel, client communications, conferences, building relationships, and establishing trust with surgeons and medical device professionals who use Lazarus 3D solutions every day.
As a primary connection to the healthcare professionals who rely on our products to improve surgical skills, operative excellence, patient safety and outcomes, you are the face and voice of our solutions. You promote our Pre-Sure and Skill-Sure products to meet customers’ clinical and marketing needs. You enjoy working with surgical teams across a multitude of specialties, including but not limited to urology, cardiothoracic, neurosurgery, general surgery, orthopedics and pediatrics. The freedom to conquer and dominate the medical education and patient-specific surgical rehearsals market without the dreadful red tape of a traditional corporation brings you joy and inspires you every day.
We are seeking individuals who take personal accountability for achieving results and can “win” at each stage of the customer's buying process. Are you ready to become a subject matter authority in our Skill-Sure and Pre-Sure portfolio? This position requires an extensive background and experience in medical device sales, medical education and simulation, working closely with healthcare administrators and the VAC for new vendor onboarding and marketing. Lazarus 3D highly values quality, precision, and clear communication. This position’s roles and responsibilities are further outlined below:
Core responsibilities include-
- Navigate the fuzzy front end of new sales development in a startup environment
- Sales of Pre-Sure and Skill-Sure aligned with corporate goals
- Catalyze revenue growth and expand market share
- Maintaining on-label usage of Pre-Sure through presence in the surgical suite and in-servicing
- Support market development, marketing, and product positioning to drive corporate goals
- Successfully build and maintain new and existing client relationships
- Travel to hospitals, operating rooms, residency training programs, CME courses, partners’ sites, and trade shows. Travel time is estimated at 15-20%
- Support strategy and coordination for trade shows, conferences, and other events
- Coordinate with R&D, clinical, and production teams to strategically develop new products
- Retain and build customer relations for customer success
- Embrace Lazarus 3D’s core values
- Comply with Lazarus 3D’s Quality System
- Other duties as assigned
What you’ll bring:
- 10+ years of experience in sales and marketing for medical devices and medical education
- Experience in market development, product positioning, new lead generation, KOL management, and customer relations, especially in the following areas: urology, cardiothoracic, neurosurgery, orthopedics, pediatrics, and transplant medicine
- Extensive experience working with Medical Schools, Medical Device Companies, Health Systems and Hospitals, Surgeons and Healthcare Administration
- New Product launch experience preferred ideally in a startup environment
Key Responsibilities:
- Execute sales strategy including both upstream and downstream marketing
- Manage budgets, forecasting, pricing, and long-term strategy
- Build and maintain relationships with key clients and deliver exceptional value
- Organize trade shows, events and activities to support corporate goals and continuous growth
- Expand utilization of Lazarus 3D products across new verticals
- Provide and present key sales metrics to Executive Management
- This position is full-time and on-site preferred, open to remote
Additional Abilities:
- Ambitious, self-driven, self-motivated
- Excellent customer relation skills, able to work well at all levels in the organization
- Ability to multitask and work independently
- Ability to work under tight deadlines
- Experience in cold outreach
- Expertise in operate CRM systems
- Extensive network in the medical device space, including knowledge of companies, products, trends, and key players, is strongly preferred
- Expertise in transplant, cardiothoracic, neurosurgery, orthopedics, and pediatrics procedures is strongly desired.
Must have a valid driver’s license and passport; must have complete vaccination records in compliance with HIPAA for OR on-site visits.
Applications should be emailed to lori@laz3d.com. CV should be titled in the following format: “CV_CL_Laz3D_Firstname_LastName_2024”. A 1-page cover letter is required - CV and cover letter should be combined into a single document. Applications submitted without a cover letter will not be reviewed. Applicants typically receive a response within 48 hours (2 business days) post-submission.
Lazarus 3D is an Equal Opportunity Employer; Employment with Lazarus 3D is governed on the basis of merit, competence and qualifications and will not be influenced in any manner by race, color, religion, gender, national origin/ethnicity, veteran status, disability status, age, sexual orientation, gender identity, marital status, mental or physical disability or any other legally protected status.
Candidates must be US citizens/Permanent residents and must be eligible to work in the United States.
Equal Employment Opportunity:
Lazarus 3D is an equal-opportunity employer. Lazarus 3D evaluates qualified applicants without regard to race, color, religion, gender, national origin, age, sexual orientation, gender identity or expression, protected veteran status, disability/handicap status or any other legally protected characteristic.
Reasonable Accommodations:
Lazarus 3D is committed to working with and providing reasonable accommodations to individuals with disabilities globally. If, because of a medical condition or disability, you need a reasonable accommodation for any part of the application or interview process, please contact us and let us know the nature of your request along with your contact information.
Recruitment Fraud Notice:
Lazarus 3D has discovered incidents of employment scams, where fraudulent parties pose as Lazarus 3D employees, recruiters, or other agents, and engage with online job seekers in an attempt to steal personal and/or financial information. Caution should be exercised to protect yourself from recruitment fraud.
Address:
620 Applegate St, Philomath, OR 97370
Appendix A: Compensation
Base Compensation:
We understand compensation is an important factor as you consider the next step in your career. At Lazarus 3D, we are committed to equitable pay for all employees, and we strive to be more transparent with our pay practices. The estimated base salary for this position is $75,000 to $85,000 base annually for new hires. Individual pay is based on skills and expertise, experience, and other relevant factors. For questions about this, our pay philosophy, and available benefits, please speak to the hiring manager if you decide to apply and are selected for an interview.
Commission:
A 10% commission will be provided for all sales of Pre-Sure and Skill-Sure driven and managed by the employee.
Stock Options:
To align the Product Specialist's interests with the company's growth, Lazarus 3D is open to negotiations on stock options at 6 months post-hiring. Stock options will follow the standard one-year cliff and 4-year vesting schedule.
Benefits Package:
Lazarus 3D provides 50% contribution to a comprehensive health, dental and vision insurance plan, paid time off, holiday pay, retirement plans through the state of Oregon (401k) for Oregon residents, free on-site parking, and other benefits such as discounts to gym memberships, pet insurance, homeowners/renters insurance, etc. to all FTE’s at Lazarus 3D.
Other Considerations:
Relocation assistance may be applicable for this position.
Associate Research Scientist
General: Full-Time, Non-Exempt Employee
Job Category: R&D Innovation
Base Salary: $65,000- $85,000, based on experience
Overview of Lazarus 3D: Surgeon skill and experience are the biggest determinants of patient outcomes. Unfortunately, surgeons learn by operating on patients and sometimes they make costly mistakes. Lazarus 3D’s Pre-Sure platform allows surgeons to rehearse every surgery ahead of time on a replica of their patients’ organs. This unparalleled opportunity to exercise their surgical plan on Pre-Sure is game-changing in surgical care as it can transform patient safety and outcomes. Lazarus 3D’s Skill-Sure platform delivers world-class solutions to healthcare professionals interested in reducing the learning curve, innovation and adoption of new medical devices. Pre-Sure and Skill-Sure models mimic soft, realistic textures of real organs and provide a unique hands-on operative experience.
Our mission is to help doctors operate with confidence: Lazarus 3D is driven by a singular mission: to enhance patient safety and outcomes through tailored solutions that empower surgeons to operate with unwavering confidence. Our vision is ambitious yet resolute—to eradicate surgical errors, ensuring that every patient receives the best care possible.
Join us at Lazarus 3D as we redefine the boundaries of surgical precision and pave the way for a future where impeccable care is the standard.
Your Role at Lazarus 3D
Lazarus 3D is seeking a highly motivated and detail-oriented Associate Research Scientist with a background in chemistry, material science, or a related field to join our team. The Associate Research Scientist will perform the duties described below and demonstrate initiative to take actions necessary to assist in R&D and to complete assignments thoroughly by verifying their own work as defined/outlined by the Principal Scientist. The Associate Research Scientist will help with the development of new material formulations and conduct qualitative and quantitative analyses of various materials that will ultimately be used to produce realistic, human tissue-simulating models and medical devices. The selected candidate will oversee all laboratory functions, including equipment maintenance, SOP development, inventory management, safety compliance, data organization, and collaborative communication. They will also drive continual improvement initiatives and ensure emergency preparedness, maintaining a high standard of efficiency, safety, and productivity in the lab.
Qualifications and Critical Skills/Expertise:
- Education minimum requirement: Graduate degree or equivalent with work experience in the fields of Chemistry, Chemical Engineering, Biomedical Engineering, Material Science and Engineering, or a related scientific discipline
- Candidate should have a solid knowledge of mechanics and deformation of soft-tissue polymers
- Experience in design and experimental operations, as well as hands-on experience with mechanical hardware is expected
- Working experience in tensile testing preferred
- Familiarity with polymers/elastomers, CAD, and prior work experience in a medical device R&D organization will be plus
- Research experience in polymers and formulation development; must have experience working in a chemicals/materials/biomedical lab, be familiar with basic synthetic (polymers or small molecules) chemistry techniques, and have hands-on experience using analytical instruments
- Knowledge of materials characterization tools (such as rheometer, Instron, spectroscopic methods, DSC, GPC, etc.) and interpreting resultant data
- Experience managing and maintaining a chemical laboratory
- Ability to install, troubleshoot, and maintain various laboratory equipment
- Ability to conduct research project activities independently
- Ability to plan project activities and meet timelines
- Must have good verbal and written communication skills
- Must have good computer skills including knowledge of Word and Excel
- Knowledge of experimental design and statistical tools
- Knowledge of GMP, GLP, or ISO standards preferred
- A collaborative and positive attitude
What you’ll do:
- Support our vision and mission: To improve patient safety and surgical outcomes
- Possess strong research acumen, including conducting thorough literature review, analyzing relevant materials, and synthesizing findings to inform innovation and development
- Set up, maintain, and manage a clean lab environment to provide a conducive workspace for experimentation and ensure adherence to safety protocols. This includes implementing effective inventory management systems and waste disposal procedures
- Conduct experiments to characterize material properties, summarize and analyze data, interpret results, and draw conclusions.
- Develop new test capabilities, test procedures, and other research on mechanical properties of materials to continually enhance our understanding and contribute to innovative solutions
- Work with biological specimens (e.g. animal tissues) for lab-scale testing
- Present scientific observations effectively to the R&D team, facilitating collaborative discussions
- Adhere to protocols and quality standards to ensure experimental consistency
- Identify, qualify, calibrate, document, and maintain lab equipment to optimize performance and accuracy. Documentation should include equipment manuals, maintenance logs, and calibration records
- Maintain a meticulously detailed lab notebook to accurately document experimental procedures, observations, and results, ensuring thoroughness and scientific integrity
- Assist in drafting various documents, including grant proposals, work instructions, and other technical documents as needed
- Experience in sterile technique/autoclave is a plus
- Learn and adapt quickly to changing needs and demands of a fast-growing startup
- Willingness to grow and expand your skill set
- Troubleshoot and identify technical alternatives
- Perform multiple tasks simultaneously and deliver high-quality results
- Responsible for timely project completion
- Take accountability and ownership of your responsibilities
- Work independently, anticipating workflow while coordinating activities
- Embrace Lazarus 3D’s core values
- Comply with Lazarus 3D’s Quality System
- Other duties as assigned
Equipment:
Mechanical testing equipment like Instron or similar is preferred. Surgical tools, including scalpels, rongeurs, suturing instruments, retractors, etc. is a plus.
Working Environment & Physical Requirements:
Approximately 80% of the time will involve work in the lab and 20% administrative tasks. This position involves duties in areas requiring PPE (including gloves and safety glasses). Travel is not routinely needed to perform essential job duties.
While performing the duties of this job, the employee will have limited exposure to biologics and occasional exposure to harsh chemicals.
Applications should be emailed to lori@laz3d.com. CV should be titled in the following format: “CV_CL_Laz3D_Firstname_LastName_2024”. A 1-page cover letter is required - CV and cover letter should be combined into a single document. Applications submitted without a cover letter will not be reviewed. Applicants typically receive a response within 48 hours (2 business days) post-submission.
Lazarus 3D is an Equal Opportunity Employer; Employment with Lazarus 3D is governed on the basis of merit, competence and qualifications and will not be influenced in any manner by race, color, religion, gender, national origin/ethnicity, veteran status, disability status, age, sexual orientation, gender identity, marital status, mental or physical disability or any other legally protected status.
Candidates must be US citizens/Permanent residents and must be eligible to work in the United States.
Equal Employment Opportunity:
Lazarus 3D is an equal-opportunity employer. Lazarus 3D evaluates qualified applicants without regard to race, color, religion, gender, national origin, age, sexual orientation, gender identity or expression, protected veteran status, disability/handicap status or any other legally protected characteristic.
Reasonable Accommodations:
Lazarus 3D is committed to working with and providing reasonable accommodations to individuals with disabilities globally. If, because of a medical condition or disability, you need a reasonable accommodation for any part of the application or interview process, please contact us and let us know the nature of your request along with your contact information.
Recruitment Fraud Notice:
Lazarus 3D has discovered incidents of employment scams, where fraudulent parties pose as Lazarus 3D employees, recruiters, or other agents, and engage with online job seekers in an attempt to steal personal and/or financial information. Caution should be exercised to protect yourself from recruitment fraud.
Address:
620 Applegate St, Philomath, OR 97370
Other Considerations:
Relocation assistance may be applicable for this position.
DISCLAIMER
Lazarus 3D is an Equal Opportunity Employer; Employment with Lazarus 3D is governed on the basis of merit, competence and qualifications and will not be influenced in any manner by race, color, religion, gender, national origin/ethnicity, veteran status, disability status, age, sexual orientation, gender identity, marital status, mental or physical disability or any other legally protected status. Candidates must be US citizens/Permanent residents and be eligible to work in the United States.